Dr. Kristin Kirschbaum definitely found her passion when it comes to crystallography. The director of the Instrumentation Center in Bowman-Oddy Laboratories and graduate student Kelly Lambright recently solved the crystal structures of huge gold nanoparticles, which were published in Science and Science Advances.
“It is an achievement and an honor for The University of Toledo in general and crystallography at UT in particular,” Kirschbaum said of the work that appeared in the top scientific journals. “Crystallography at UT has had a great reputation over many decades, under the leadership of Dr. A. Alan Pinkerton, and we are proud to have worked over the years with high school, undergraduate and graduate students. I am particularly delighted that a large part of this work was done by one of our graduate students from the chemistry department, Kelly J. Lambright.
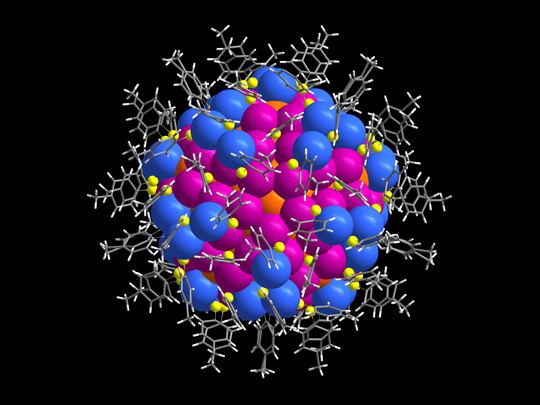
This is the UT team’s crystallographic analysis of the nanoparticle consisting of 246 gold atoms that was published in Science. The orange, blue and pink spheres are the gold atoms, yellow is sulfur, and grey and white are the carbon and hydrogen protective organic layer.
The recent article in Science focuses on crystallography solving the atomic structure of a Au246 nanoparticle revealing the three-dimensional arrangement of all 246 gold atoms and the protective organic shell. This kind of information can only be determined by crystallography.
“First, you start with beautiful crystals,” said Kirschbaum cheerfully, isolating a single crystal sample, and placing it inside a diffractometer. The Instrumentation Center is home to five of these instruments, and it’s common for the lab to receive crystals from around the world to analyze; requests from places such as Iran and India, but also national collaborators from Duke, University of Houston, Sloan-Kettering Cancer Center and Carnegie Mellon. The Carnegie Mellon collaborator Dr. Rongchao Jin, an internationally recognized expert in nanoparticles, provided the crystal of Au246 nanoparticles.
The crystal is then exposed to an x-ray beam, which it diffracts in a way that is unique to that particular compound. These reflections are collected and analyzed to find the 3D structure of the compound.“The importance of these structural results lies in the fact that all of the medical, electronic and optical properties differ depending on the size and the structure of the nanoparticles. Certain nanoparticles are known to enhance cancer radiotherapy, others assist in early tumor detection or are used in solar cells and nano-electronics,” Kirschbaum explained. “The secret of why some nanoparticles have certain properties and others do not is in the 3D arrangement of the atoms. The publication in Science is based on the crystal structure analysis of a novel nanoparticle. It is the largest gold nanoparticle so far whose structure has been determined by crystallography. The foremost largest crystallographically analyzed gold nanoparticle consisted of 133 gold atoms and was also just recently structurally characterized at UT and published in 2015 in Science Advances.”
Kirschbaum went on to say how the research done at UT can have tremendous applications: “Biomedical applications include sensing of important biomolecules — for example, glucose — radio-sensitizing for cancer radiotherapy and early tumor diagnosis. Knowing the arrangement of the atoms allows understanding and predicting the properties of these nanoparticles. Scientists can then design novel nanoparticles with a certain structure and desired properties.”